COVAXIN, India’s Indigenous vaccine developed by Bharat Biotech unlike existing vaccines against , it has been found that the inactivated virus vaccine induces robust immune memory to SARS-CoV-2 and multiple variants of concern. It also targets not just mutations in spike proteins but also multiple proteins, thereby offering protection against alpha, beta, gamma, kappa and delta variants according a study conducted by National Institute of Immunology.
These findings establish insight into the attributes of BBV152-elicited immune memory, and has implication for future vaccine development, guidance for use of inactivated virus vaccine, and booster immunization.
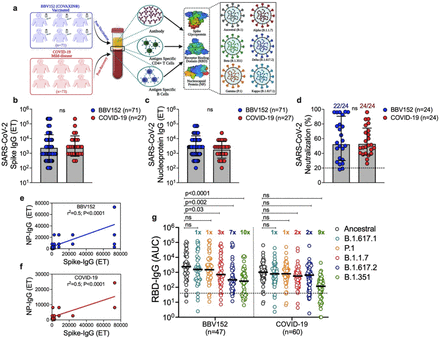
Furthermore, the protection offered by the indigenous vaccine is generated through memory cells (Tcell, B cell), which is long-term and is not affected by waning of antibodies. This has been concluded by a group of scientists in the first ever preprint study on COVAXIN led by the National Institute of Immunology.
COVAXIN offers robust protection against variants of concerns
“We studied in a cohort for six months where Covaxin induced good memory response. So we are confident that till six months of full immunisation, no booster dose is required. A follow up of this cohort thereafter, will help us to understand if we really need booster shots or not,” said Dr Nimesh Gupta, corresponding author of the study at NII.
“The best protection was found against alpha then delta and the least was against beta variants,” he added.
The study by Nii found that covaxin induces memory B cells, similar to natural infection, which are impacted by virus variants in the same order as antibodies
The immune response induced by the vaccine was found to be similar to the natural infection. “We have the whole virus and our immune system mounts the responses to all the proteins of the virus. The impact of this vaccine may be similar to the natural infection where it allows our immune system to mount the memory responses against multiple
proteins.
The benefit is that if a huge variant comes from a mutation in the spike protein, which may escape the vaccine only targeting spike proteins, in this vaccine there are some supplementary protective responses from responses against other proteins,” said Dr Gupta.
Also, the vaccine, in low doses, can be tested on the paediatric population. “We do not need a high quantity of immune responses for Covid but they should be optimum. In children, low doses can be protective,” said the scientist.
Researchers believe that the memory generation is mainly due to the adjuvant in the vaccine which has been used for the first time. “Whole virus has a response against multiple proteins so that you can be completely protected against variants. And this adjuvant is generating immune responses- -which is very close to the nature of natural infections,” said Dr Gupta.
Further, the vaccine induces memory B cells, similar to natural infection, which are impacted by virus variants in the same order as antibodies. The vaccine further induced antigen-specific functionally potent multi-cytokine expressing CD4+ T cells in ∼85% of the subjects, targeting spike and nucleoprotein of SARS-CoV-2. Marginal ∼1.3 fold-reduction was observed in vaccine-induced CD4+ T cells against the beta variant, with no significant impact of the alpha and the delta variants. The antigen-specific CD4+ T cells were populated in the central memory compartment and persisted up to 6 months of vaccination. Importantly the vaccine generated Tfh cells that are endowed with B cell help potential, similar to the Tfh cells induced after natural infection. Altogether, these findings establish that the inactivated virus vaccine BBV152 induces robust immune memory to SARS-CoV-2 and variants of concern, which persist for at least 6 months after vaccination
The study was conducted in collaboration with AIIMS New Delhi, Maulana Azad Medical College and Lok Nayak hospital, Delhi, Center for Infectious Disease and Vaccine Research, La Jolla Institute for Immunology, California, Division of Infectious Diseases and Global Public Health, University of California, San Diego, Translational Health Science and Technology Institute, Faridabad and ESIC Medical College and Hospital, Faridabad.





![[New Data] Moderna Vaccine Jabs has high heart inflammations risk](https://thenewsfacts.com/wp-content/uploads/2021/10/moderna-vaccine-could-cause-heart-inflamation-thenewsfacts.com_-1.jpg)
